NLP Generated High Complexity Combination Therapy for Pancreatic Cancer
PI-Technion, Assoc. Prof. Yosi Shamay, PhD PI-Rambam, Prof. Irit Ben Aharon, MD
Objectives and clinical need Pancreatic ductal adenocarcinoma (PDAC) is among the most lethal malignancies, with extremely poor long-term survival. One major reason for therapeutic failure is the dense fibrotic tumor microenvironment characteristic of PDAC. The extensive extracellular matrix surrounding pancreatic tumors creates a physical barrier that limits the penetration of anticancer drugs into the tumor tissue, reducing the effectiveness of otherwise potent therapies.
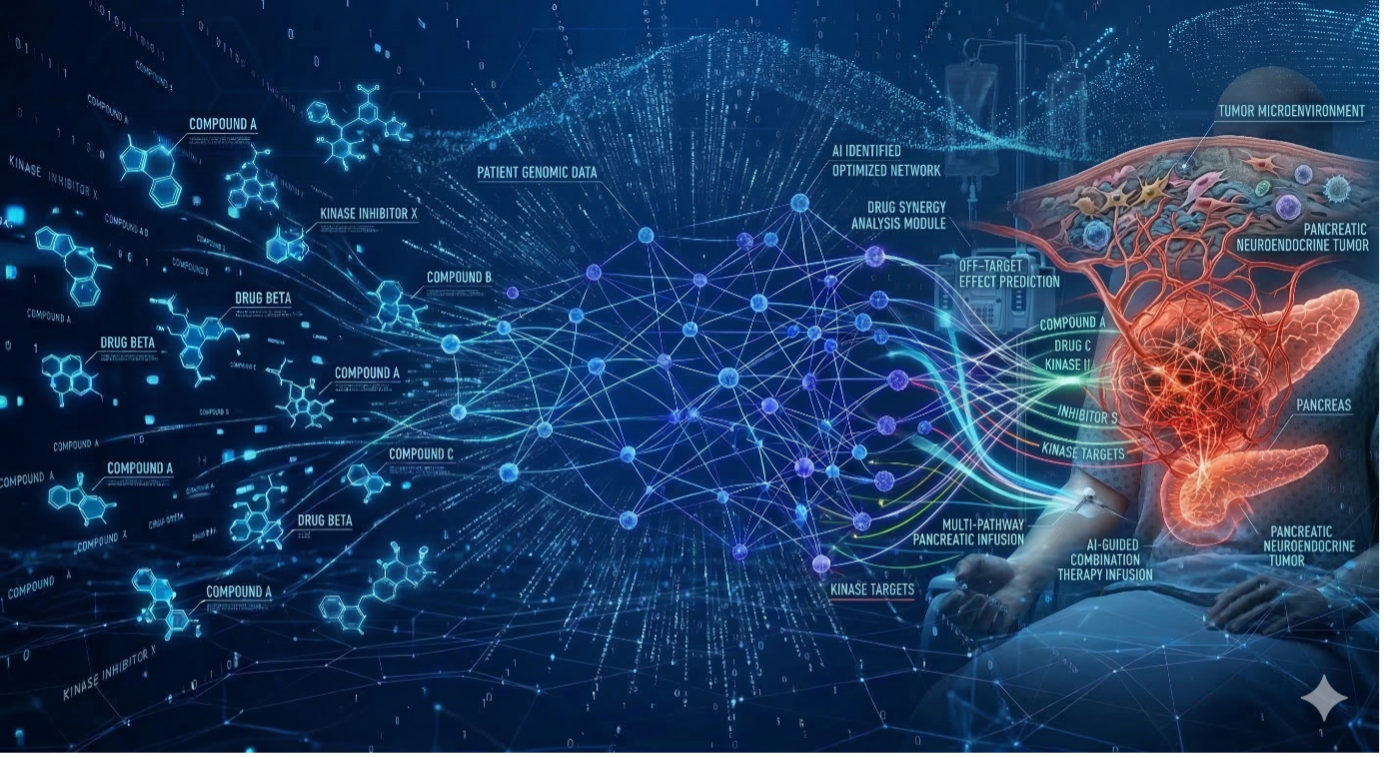
This project aims to address this challenge by developing a computational framework for designing high-complexity combination therapies (HCCT) that simultaneously target tumor cells and the surrounding stroma. By integrating artificial intelligence, large-scale literature mining, and clinical knowledge, we seek to systematically identify drug combinations capable of improving drug penetration and therapeutic response in pancreatic cancer.
Databases and cohorts
To identify candidate therapies, we constructed a knowledge base of drugs, molecular targets, and biological activities related to pancreatic cancer fibrosis and stromal modulation. Biomedical literature was systematically mined using the SPIKE search engine and SPIKE-KBC platform, enabling the automated extraction of relationships between drugs, genes, pathways, and anti-fibrotic activities.
The resulting knowledge base integrates FDA-approved drugs with potential anti-stromal activity, including agents such as losartan, metformin, nintedanib, pirfenidone, and rapamycin. In parallel, we are initiating a retrospective analysis of electronic medical records (EMR) from PDAC patients treated at Rambam Health Care Campus to evaluate the real-world clinical impact of these drugs when used alongside standard therapies.
Research results
Using the literature-derived knowledge base, we developed a computational Plan Builder system that generates candidate HCCT treatment plans. The algorithm analyzes evidence from biomedical literature and constructs networks of interacting drugs based on mechanistic relationships, potential synergy, and clinical feasibility.
The framework generates treatment strategies ranging from four to seven drugs and evaluates them using a graph-based scoring method that balances evidence strength, number of treatment elements, and interconnectivity between drugs. High-scoring plans typically integrate standard chemotherapies such as gemcitabine with anti-stromal agents and targeted therapies.
The system also translates drug networks into temporally coordinated treatment schedules, demonstrating the feasibility of implementing complex multi-drug regimens in clinical protocols. Ongoing work focuses on validating these computational predictions through clinical data analysis and expert oncologist evaluation, with the goal of identifying promising treatment strategies for future clinical trials in pancreatic cancer.



